 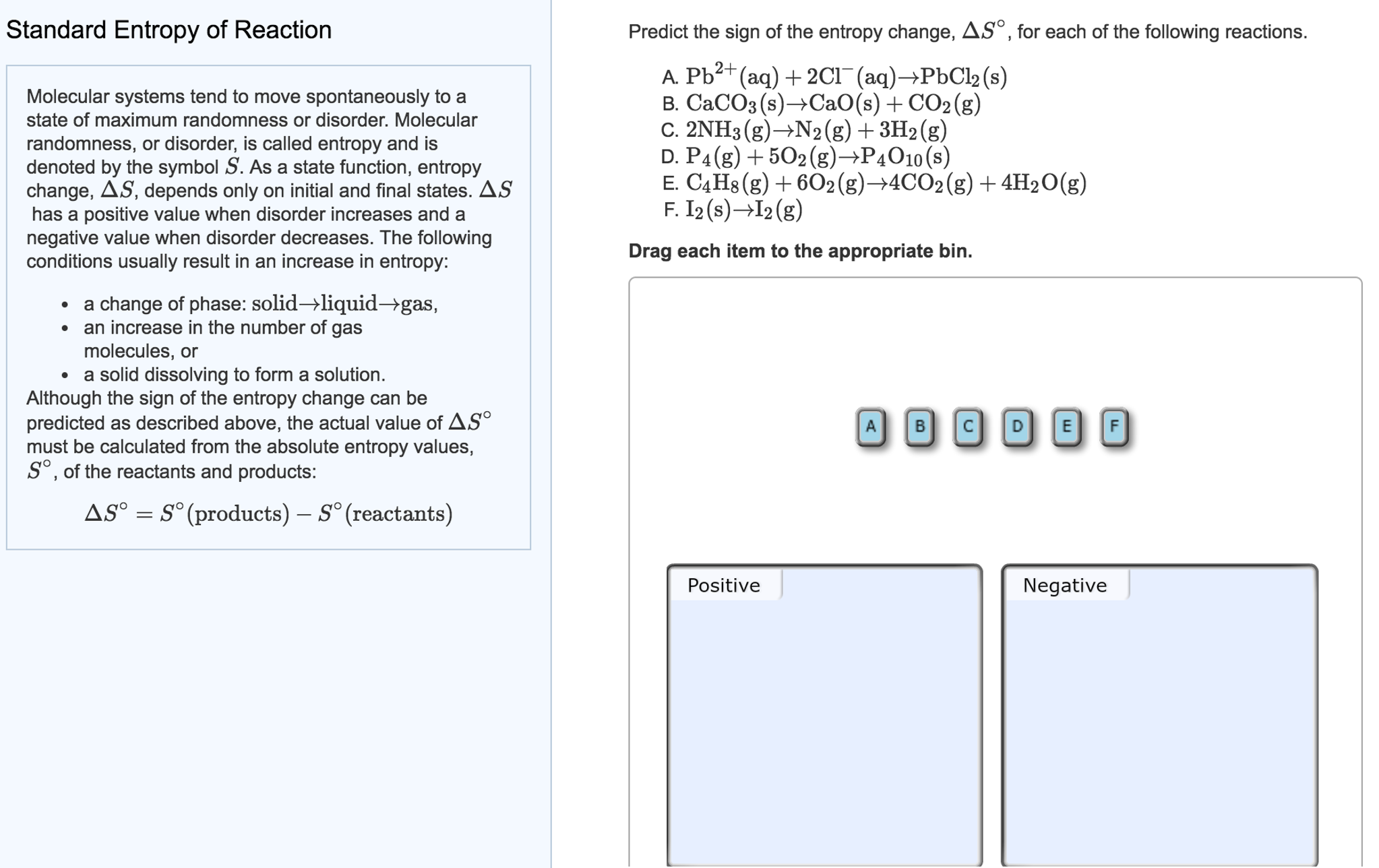
In my judgement, devising a method to cause the heat flow to occur gradually and quantitatively measuring the heat flow directly would be the hard part. See also Water Boiling points at high pressure. The standard entropy change for the reaction4Fe(s)+3O2(g)2Fe2O3(s) is 550JK1 at 298K.Given : The standard enthalpy change for the reaction is 165 kJ. See Water and Heavy Water - thermodynamic properties. Earhart 2 of 2 All standard state, 25 C and 1 bar (written to 1 decimal place). Of course, in many cases, it is much easier to calculate the entropy change (based on identifiable reversible paths) than it is to measure. The figures and tables below shows how water enthalpy and entropy changes with temperature (C and F) at water saturation pressure (which for practicle use, gives the same result as atmospheric pressure at temperatures < 100 C (212F)). The standard entropy change for the reaction 4Fes + 3O2g 2Fe2O3s is 550 J K1 at 298 K.Given: The standard enthalpy change for the reaction is 165 kJ. In some specific cases it could be done indirectly, such as in the isothermal quasistatic expansion of a gas (say in contact with a heat bath), where the measured amount of work (determined say by gradually removing small weights from a piston) would be equal to the amount of heat added. The hard part would be measuring the amount of heat flow. Standard entropy is a measure of the entropy of one mole of a substance at standard atmospheric pressure (1 atmosphere) and temperature (298 Kelvin). The experiment has to be carried in as close to reversible conditions as possible, since, in the real world, there is no perfectly reversible path. So you have to identify a path that is easy to implement, and for which the heat flow can be measured easily (say by phase change in a reservoir). 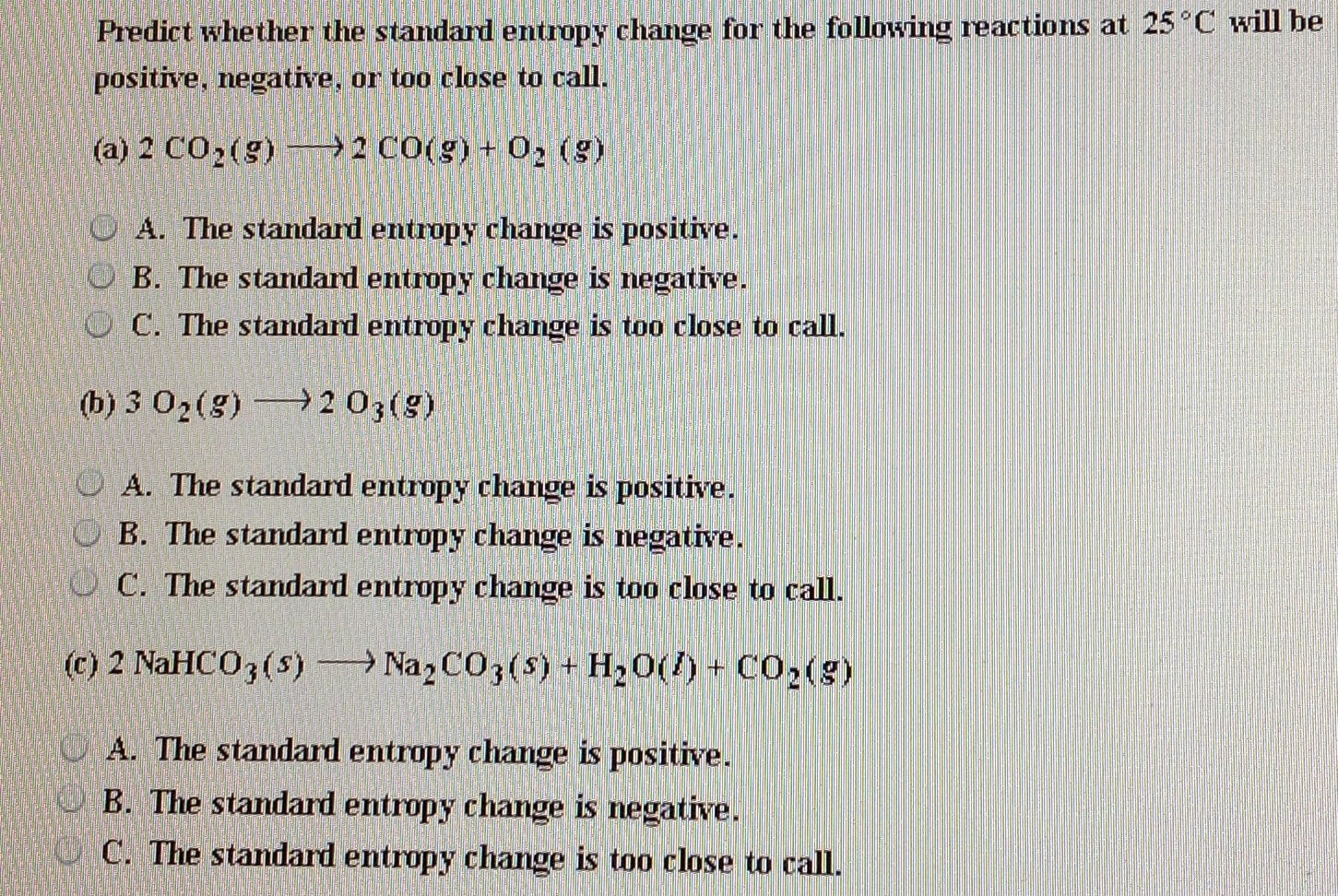
Any convenient reversible path will do, since the integral of dq/T is the same for all reversible paths. To do so, one needs to devise (dream up) a reversible path between the initial and final states. The entropy change between two thermodynamic equilibrium states of a system can definitely be directly measured experimentally. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
